CAPR Stock Sinks After-Hours: Capricor Launches Legal Fight Over DMD Therapy Rollout Ahead Of FDA Review

- Capricor alleged that NS Pharma failed to prepare for Deramiocel’s commercial rollout and refused to fix pricing flaws in the commercialization agreement.
- The company claims that the current pricing structure could make Deramiocel commercially unviable under Medicare reimbursement rules.
- Capricor said that it tried to renegotiate pricing terms, but alleged that NS Pharma “refused to compromise.”
Shares of Capricor Therapeutics, Inc. (CAPR) plunged 13% in after-hours trading on Thursday after the company sued its commercial partner over the launch of its Duchenne muscular dystrophy (DMD) therapy Deramiocel, setting off a legal fight just months before a key FDA decision.
CAPR stock ended over 1% lower at $33.94 on Monday. However, shares are on track to post their best week in three weeks.
Capricor Sues NS Pharma Over Deramiocel
Capricor filed a lawsuit against Nippon Shinyaku and its U.S. unit NS Pharma, accusing the companies of failing to prepare for the commercial rollout of Deramiocel and refusing to resolve pricing flaws within the companies’ commercialization and distribution agreement.
Deramiocel is a cell therapy derived from donor heart cells and can reduce inflammation caused by muscle damage in DMD patients. Capricor alleged that the dispute now threatens patient access to the therapy despite Deramiocel remaining under FDA review with a Priority Review PDUFA target action date of Aug. 22.
According to the lawsuit, Capricor believes its current commercialization agreement with NS Pharma could make Deramiocel commercially unviable once Medicare reimbursement rules take effect. The company alleged that hospitals, treatment centers, and distributors could lose money administering the therapy to insured patients under the existing pricing structure, potentially limiting patient access even if the drug wins FDA approval.
Capricor said it repeatedly tried to renegotiate the pricing terms but claimed NS Pharma “refused to compromise.” The lawsuit also alleges that NS Pharma later proposed a new structure that would have given the distributor greater control over pricing and reimbursement decisions. Capricor argued that the agreement had become “commercially impossible” to operate under its current terms.
CAPR Alleges Launch Delays
The lawsuit also alleges that NS Pharma slowed or stopped launch preparations for Deramiocel after the FDA issued a Complete Response Letter (CRL) last year. Capricor claims that the company entered a “pencils down” mode that disrupted launch activities, including pricing strategy, market-access planning and distribution ahead of a potential approval.
Capricor further alleged that NS Pharma failed to build the operational systems needed for a commercial rollout while the therapy remained under FDA review. The company is asking the court to allow it to pursue an alternative path to commercialize Deramiocel if approved, and to declare that NS Pharma breached parts of the agreement.
“I have spent nearly two decades building Capricor with one goal in mind: making Deramiocel available to treat these boys,” Linda Marban, CEO of Capricor, said in a statement. “I know what every additional month of delay costs them, because I know what is happening inside their muscles when they cannot be treated. There is no version of this case in which I am willing to watch NS Pharma’s inaction take that away from them.”
CAPR Investors Watch FDA Closely
The legal dispute comes as investors closely monitor the FDA review of Deramiocel and broader leadership changes within the agency’s biologics division. The FDA resumed review of Deramiocel earlier this year, following the July CRL. Capricor’s resubmission to the FDA included updated Phase 3 Hope-3 trial data showing that the therapy slowed the worsening of arm and upper-body function, as well as the progression of heart disease, compared with placebo.
CAPR stock drew heightened retail chatter recently after reports that Houman Hemmati, a former Capricor executive, is among the candidates being interviewed by FDA Commissioner Marty Makary to lead the FDA’s Center for Biologics Evaluation and Research, or CBER.
CBER oversees approvals for vaccines and advanced cell and gene therapies, including rare-disease treatments such as Deramiocel. The leadership change comes after the recent departure of former CBER director Vinay Prasad, whose tenure drew criticism over regulatory decisions involving rare-disease and gene-therapy companies, including Sarepta Therapeutics, UniQure and Regenxbio.
How Do Retail Traders Feel About CAPR?
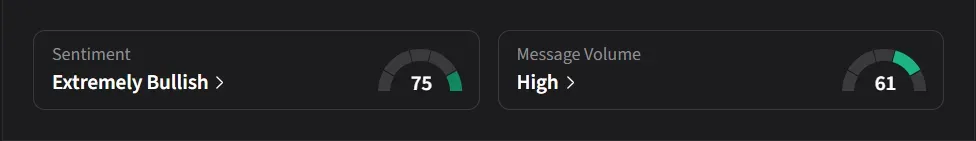
On Stocktwits, retail sentiment was ‘extremely bullish’ amid ‘high’ message volume.
One user said, “Approval could come earlier in nice bold print in the legal filing; Cant see them not winning this suit.”
Another user said, “Don't forget that distribution is going to be super simple in the US. Parents will line up with their boys to get Deramiocel. NS screwed up and Capricor is taking distribution over IMO.”
CAPR stock has risen 18% over the past year.
For updates and corrections, email newsroom[at]stocktwits[dot]com.
Read Next: GILD Stock Slips After Hours On Sharp Earnings Guidance Hit From Massive M&A Spree

/filters:format(webp)https://news.stocktwits-cdn.com/large_Trending_stock_resized_may_jpg_bc23339ae7.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/vivekkrishnanphotography_58_jpg_0e45f66a62.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2263085688_1_jpg_87213df74e.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Chinmay_Rautmare_Photo_jpeg_96dd37efd0.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Musk_Space_X_jpg_28cee07c59.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/Prabhjote_DP_67623a9828.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2196132812_jpg_086f367c0b.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_88732906_jpg_e18350eee2.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/Rounak_Author_Image_7607005b05.png)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Salesforce_resized_jpg_f6c36dbac3.webp)