Advertisement|Remove ads.
IBRX Stock Slips Overnight: Founder Teases Turkey Push, Says AI Cancer Cell Robot Is ‘Almost There’

- Founder Patrick Soon-Shiong hinted at expansion plans in Turkey, saying on X that “Global expansion” is underway.
- Soon-Shiong also revealed that ImmunityBio’s AI-driven NK-cell manufacturing robot is nearing completion.
- The AI-powered manufacturing platform, internally called “Leonardo,” can mass-produce natural killer immune cells.
Shares of ImmunityBio, Inc. (IBRX) declined marginally in overnight trading on Tuesday even after founder Patrick Soon-Shiong teased expansion plans in Turkey and revealed that the company’s AI-driven robot for mass-producing cancer-killing immune cells is “almost there.”
IBRX stock fell 5% on Monday to end at $8.11.
IBRX Turkey Buzz, AI Robot Nears Launch
“Turkey is an amazing country with such enthusiasm. Global expansion happening!” Soon-Shiong said on X, adding fresh momentum to investor speculation that ImmunityBio is accelerating its international commercialization strategy around Anktiva and next-gen cell therapies.
In a separate post from Italy, Soon-Shiong said he was inspecting the company’s AI-driven manufacturing robot as the system nears completion. “The year of the fire horse. Now in Italy and inspecting our AI-driven robot nearing completion,” he said. “A good sign of what is to come!! World Bank of NK cells.”
Soon-Shiong later posted another image showing himself inside a manufacturing facility surrounded by industrial robotic arms. “Almost there! We will transform cell therapy at scale. Beautifully made robot,” he said.
IBRX Bets Big On AI Cell Manufacturing
The latest updates build on ImmunityBio’s broader push into automated immune-cell manufacturing and advanced cancer therapies. Last week, Soon-Shiong revealed that its cell manufacturing robot had entered production and said that delivery was expected this month. He called the platform an AI and machine-learning-driven manufacturing system that can mass-produce natural killer, or NK, immune cells at industrial scale.
The robot, internally known as “Leonardo,” is expected to automate the engineering and expansion of NK cells for future off-the-shelf CAR-NK therapies. Soon-Shiong has repeatedly outlined ambitions to create a global “World Bank of NK cells.”
Earlier this year, he said ImmunityBio had already produced billions of NK cells and cryopreserved samples from more than 60 healthy donors and cancer patients. The company believes the automated manufacturing platform could eventually allow the production of trillions of NK cells, while helping decentralize cell-therapy production through installations at cancer centers globally.
Anktiva Global Rollout Gains Momentum
Soon-Shiong’s latest Turkey comments come after a series of international expansion updates for ImmunityBio’s immunotherapy platform. Recently, the biotech entrepreneur posted about meetings in Greece with Prime Minister Kyriakos Mitsotakis, Health Minister Adonis Georgiadis and Ecumenical Patriarch Bartholomew I.
“N.K. cells are God’s gift to humanity and Greece has embraced this for their people,” Soon-Shiong wrote at the time. “Global expansion continues.” Investor attention around overseas growth has intensified after ImmunityBio secured approvals for its flagship immunotherapy candidate, Anktiva, across Saudi Arabia, the UK, the EU and Macau. The company has also expanded through partnerships with Biopharma and Cigalah Healthcare in Saudi Arabia as part of a broader Middle East and North Africa strategy.
Anktiva FDA Review Remains Key Focus
Another major focus for investors remains the FDA’s ongoing review of ImmunityBio’s resubmitted supplemental biologics license application for Anktiva plus standard BCG in non-muscle-invasive bladder cancer patients with papillary tumors. The filing is backed by long-term Quilt 3.032 data, which the company said showed that 58.2% of patients remained cancer-free after 12 months while more than 80% avoided bladder-removal surgery over three years.
The FDA backdrop has become even more closely watched after reports said that U.S. President Donald Trump approved a plan to potentially fire FDA Commissioner Marty Makary amid tensions over the agency’s regulatory agenda and approval decisions.
ImmunityBio has increasingly positioned Anktiva and its broader “Immunotherapy 2.0” platform as alternatives to traditional chemotherapy and checkpoint-based oncology treatment approaches. Soon-Shiong recently criticized existing cancer treatment standards after citing data showing that many advanced lung cancer patients still fail to receive effective treatment. “Chemo. Radiation then checkpoints, then even more chemo!!” he said. “When do we learn that this can change with immunotherapy?”
How Do Retail Traders Feel About IBRX?
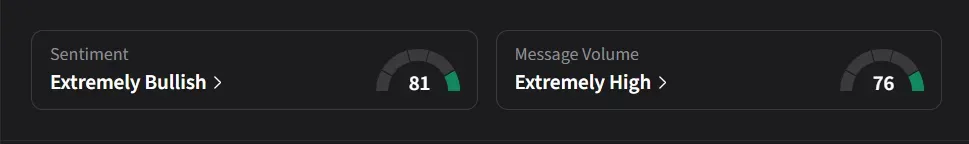
On Stocktwits, retail sentiment for IBRX was ‘extremely bullish’ amid a 332% jump in 24-hour message volumes.
One user said they had a “little dream” of a “spinoff dividend from our company with our robot manufacturing”
Another user said, “from the perspective of an international (non-US) shareholder, the more acceptance ANKTIVA receives internationally, the less important the US acceptance is.”
IBRX stock has surged 292% over the past year.
For updates and corrections, email newsroom[at]stocktwits[dot]com.
Read Next: VCX Becomes Retail’s AI Darling On SpaceX, Anthropic Hype — One Investor Warns Of 50% Crash

/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2269812042_jpg_6a69c0f17a.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/1707726834303_jpg_11f20a9aa9.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2264270294_jpg_97b0b72b9d.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Whats_App_Image_2026_05_11_at_09_45_43_1_jpeg_a08c0cf251.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Trending_stock_resized_may_jpg_bc23339ae7.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/vivekkrishnanphotography_58_jpg_0e45f66a62.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2275396018_jpg_a205b2aecb.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/Prabhjote_DP_67623a9828.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2275353529_jpg_271d3f9c9b.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Anushka_Basu_make_me_smile_in_the_picture_b92832aa_af59_4141_aacc_4180d2241ba8_1_2_png_1086e0ed8c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2238553113_jpg_02cf1c9b5e.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/Rounak_Author_Image_7607005b05.png)