Advertisement|Remove ads.
NVO Stock Pops Premarket: Novo Nordisk Unveils Fresh Wegovy Data That Could Narrow Eli Lilly’s Obesity Lead
- Novo said fast-responding patients taking Wegovy 7.2 mg lost an average 27.7% of their body weight after 72 weeks.
- The company said patients on higher-dose Wegovy reached weight-loss goals faster than those taking the standard 2.4 mg version.
- Novo added that 84% of the weight lost with Semaglutide came from reductions in body fat, while muscle strength and muscle health were largely preserved.
U.S.-listed shares of Novo Nordisk (NVO) jumped 3% in premarket trading on Tuesday after the drugmaker unveiled fresh Wegovy data showing that some patients lost nearly 28% of their body weight, adding fuel to its intensifying battle with Eli Lilly over obesity drugs.
Novo’s U.S.-listed shares closed nearly 1% higher on Monday to $46.4, recording its eighth straight session of gains.
Novo Flags Stronger Wegovy Results
The new data, presented at the European Congress on Obesity in Turkey, came from a fresh analysis of Novo’s Step Up trial, which studied a higher-dose 7.2 mg version of Wegovy. Novo said that patients who responded quickly to treatment, defined as losing at least 15% of their body weight within the first six months, went on to lose an average 27.7% of their body weight after 72 weeks.
Novo also said patients taking the higher-dose version reached weight-loss goals faster than those on the currently marketed 2.4 mg Wegovy dose. According to Novo, about 84% of the weight loss with Semaglutide was due to reductions in body fat, while muscle strength and muscle health were largely preserved.
“The analyses from STEP UP presented at ECO are really promising,” Emil Kongshoj Larsen, executive vice president and head of International Operations at Novo Nordisk, said in a statement.
Novo Expands Wegovy HD Rollout
In January, Novo said that patients taking weekly Semaglutide 7.2 mg achieved average weight loss of 20.7% over 72 weeks, compared with 17.5% for the standard 2.4 mg Wegovy dose. About one-third of patients on the higher dose lost at least 25% of their body weight. The FDA approved Wegovy HD in March, and Novo launched the stronger version in the U.S. last month.
The Step Up study included over 1,400 adults living with obesity, but without type 2 diabetes. Novo said that the higher-dose version maintained a safety and tolerability profile generally consistent with lower-dose semaglutide treatments. Novo is also seeking approval in Europe and the U.K. for a single-dose 7.2 mg Wegovy pen, with regulatory decisions expected later this year.
Novo, Lilly Obesity Race Heats Up
The latest Wegovy data comes as Novo tries to strengthen its position against Eli Lilly, whose obesity drug Zepbound has increasingly dominated the market after posting stronger weight-loss results in head-to-head studies.
On Monday, Lilly released detailed results from its Surmount-5 trial, showing that patients taking Zepbound lost an average 20.2% of their body weight over 72 weeks, compared with 13.7% for patients taking traditional Wegovy dosing. Nearly half of Zepbound patients achieved at least 20% weight loss versus about 27% for Wegovy.
Novo’s latest 27.7% figure is not directly comparable since it reflects only a subgroup of fast responders rather than the overall trial population. The update marks a sign that Novo may be starting to narrow the performance gap with Lilly by offering a more powerful Wegovy option for patients who need greater weight reduction.
Novo, Lilly Expand Battle Into Pills
The rivalry between Novo and Lilly is also spreading into oral obesity drugs. Novo launched its once-daily Wegovy pill in January, giving the company an early lead in the category. Lilly followed in April with Foundayo, its own oral obesity pill.
Early prescription tracking since Eli Lilly launched Foundayo in April has favored Novo Nordisk so far. Foundayo generated 3,707 prescriptions in its second week and 7,335 prescriptions in its fourth week, trailing the earlier launch pace of Novo’s oral Wegovy pill, according to IQVIA prescription data cited in market reports. Lilly recently said that Foundayo already has over 8,000 prescribers and over 20,000 treated patients, with more than one-third of prescribers new to oral GLP-1 therapies.
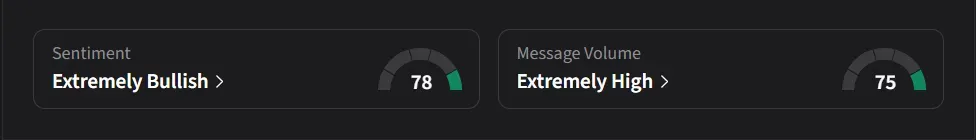
How Do Retail Traders Feel About NVO And LLY?
On Stocktwits, retail sentiment for NVO was ‘extremely bullish’ with ‘extremely high’ message volume, while sentiment toward LLY remained ‘bearish’ with ‘normal’ message activity.
One user said, “Pill efficacy doesn’t matter to the market cause it’s higher than Lilly’s “but but convenience”, so hope this one matters since it’s a much more fair comparison to Zepbound than with 2,4mg.”
Another user said NVO’s recent stock move “feels less like a dead cat bounce and more like a slow sentiment reversal after months of fear around growth, competition, reimbursement, and management concerns.”
While Novo’s U.S.-listed shares fell 27% over the past year, LLY shares jumped 33%.
For updates and corrections, email newsroom[at]stocktwits[dot]com.

/filters:format(webp)https://news.stocktwits-cdn.com/large_POET_technologies_OG_jpg_ebaa71a878.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/IMG_9209_1_d9c1acde92.jpeg)
/filters:format(webp)https://news.stocktwits-cdn.com/large_wendy_s_resized_jpg_9b298d0aee.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/shivani_photo_jpg_dd6e01afa4.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_novo_nordisk_ozempic_wegovy_jpg_786cdf3b34.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/IMG_8805_JPG_6768aaedc3.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/large_US_stocks_3e2253bcca.jpg)
/filters:format(webp)https://news.stocktwits-cdn.com/Revised_Profile_JPG_0e0afdf5e2.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Getty_Images_2211073858_jpg_993ab069bd.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_musk_jpg_06b3732249.webp)