Advertisement|Remove ads.
CAPR Stock In Focus: Ex-Exec Eyed For FDA Role Says Drug Approvals Are Not ‘Best-In-Class’ Signals

- Hemmati is reportedly being considered for a leadership role at the U.S. Food and Drug Administration’s Center for Biologics Evaluation and Research.
- The regulatory spotlight has intensified following recent FDA moves, including the fast-tracked expanded access for Revolution Medicines’ cancer drug, Daraxonrasib.
- Capricor awaits an FDA decision on Deramiocel by Aug. 22.
Shares of Capricor Therapeutics, Inc. (CAPR) drew retail attention on Monday after a former executive, eyeing a potential FDA chief role, said drug approval should only be viewed as a “building permit” rather than a sign that a therapy is the top choice.
CAPR stock declined 8% on Friday as investors tracked regulatory developments ahead of a high-stakes decision on its Duchenne muscular dystrophy (DMD) therapy, Deramiocel.
The focus increased after Houman David Hemmati, who previously served at the company and is now being considered for a leadership role at the U.S. Food and Drug Administration (FDA).
Hemmati: Approval Is Just A Safety Check
Hemmati said on X that FDA approval does not indicate that a drug is “incredible” or the best option available, but simply confirms it meets established standards for safety and efficacy. “It’s like a building permit: it simply confirms the therapy meets rigorous standards for safety and efficacy,” he said.
He said that patients can choose not to take a drug, doctors may decide not to prescribe it, and insurers may opt not to cover it if it is not appropriate, stressing that approval is only one part of a broader framework for real-world use.
“Taking away choice because a drug isn’t “absolutely perfect” means patients and physicians never even get the option to consider it. Putting patients first means preserving every possible tool—so families and doctors can decide together,” Hemmati said.
Hemmati Emerges As FDA Role Contender
Hemmati, who served as vice president of medical and clinical development at Capricor between 2015 and 2017, is reportedly among candidates being considered to lead the FDA’s Center for Biologics Evaluation and Research, which oversees gene and cell therapy approvals.
The potential leadership change follows the departure of CBER director Vinay Prasad, whose tenure drew criticism from the biotech sector over decisions affecting rare-disease therapies.
Recent regulatory decisions have also drawn focus to the agency’s approach. The FDA last week granted a “safe to proceed” letter to Revolution Medicines for expanded access to its pancreatic cancer drug Daraxonrasib, allowing treatment for patients with previously treated metastatic disease.
The agency cleared the request within two days of submission, with Commissioner Marty Makary saying that the move is a commitment to facilitate early access to therapies for serious and life-threatening conditions. Daraxonrasib, a RAS inhibitor, has received Breakthrough Therapy and Orphan Drug designations and is expected to be submitted for approval under the agency’s National Priority Voucher program.
CAPR Awaits High-Stakes DMD Decision
The developments come as Capricor awaits an FDA decision on Deramiocel, expected by Aug. 22. The therapy targets DMD, a genetic condition that causes progressive muscle weakness in children.
The FDA resumed its review in March after issuing a Complete Response Letter in July 2025 requesting additional data. Capricor’s resubmission included Phase 3 Hope-3 trial results showing a 54% slowing in upper-limb function decline and a 91% reduction in cardiac disease progression compared with placebo. Deramiocel consists of donor-derived cardiac cells to reduce inflammation from muscle degeneration in DMD.
How Do Retail Traders Feel About CAPR?
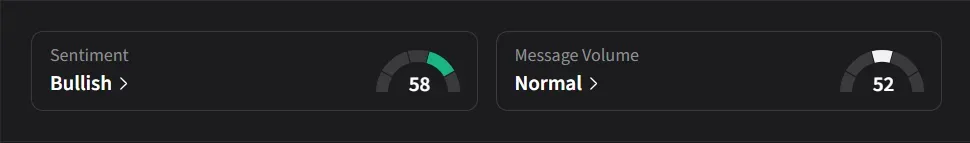
On Stocktwits, retail sentiment for CAPR was ‘bullish’ amid ‘normal’ message volume.
One user called CAPR a “Clinical-stage biotech with high risk and potential reward tied to trial success”
Another user said, “4 months max until we can finally start selling and get treatment for these boys. Time really flies since our end of feb submission”
CAPR stock has surged 143% over the past year.
For updates and corrections, email newsroom[at]stocktwits[dot]com.
Read Next: Nio Stock Jumps Overnight: EV Maker Brings Back Original Chief To Run Software Behind Its Cars

/filters:format(webp)https://news.stocktwits-cdn.com/large_tom_lee_fundstrat_bmnr_OG_jpg_94a105e116.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/Anushka_Basu_make_me_smile_in_the_picture_b92832aa_af59_4141_aacc_4180d2241ba8_1_2_png_1086e0ed8c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2246754937_jpg_b51052741c.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2159666976_jpg_d3f861259a.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2264775602_jpg_64a2d5354a.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_1925090354_jpg_f55e4049ea.webp)
/filters:format(webp)https://news.stocktwits-cdn.com/large_Getty_Images_2244288176_jpg_c3909aa8e5.webp)
/filters:format(webp)https://st-everywhere-cms-prod.s3.us-east-1.amazonaws.com/Prabhjote_DP_67623a9828.jpg)